Ethenone Synthesis From Acetone on:
[Wikipedia]
[Google]
[Amazon]
In
C2H4O2 -> C2H2O + H2O
On a laboratory scale it can be produced by the thermal decomposition of Meldrum's acid at temperatures greater than 200 °C.
 The thermal decomposition of acetic anhydride was also described.
The thermal decomposition of acetic anhydride was also described.



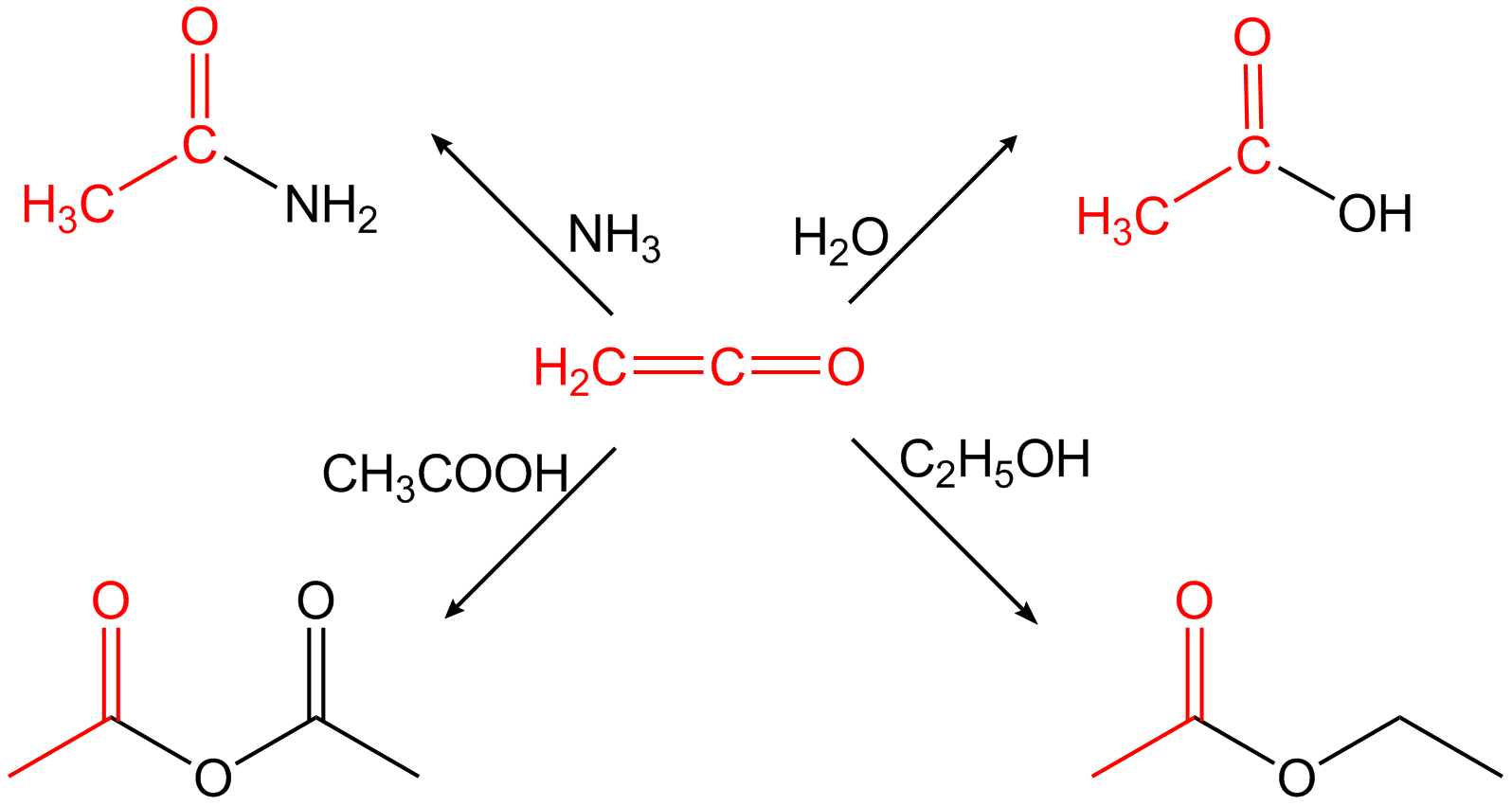 :
: Ethenone reacts with methanal in the presence of catalysts such as Lewis acids (AlCl3, ZnCl2 or BF3) to give β-propiolactone. The technically most significant use of ethenone is the synthesis of
Ethenone reacts with methanal in the presence of catalysts such as Lewis acids (AlCl3, ZnCl2 or BF3) to give β-propiolactone. The technically most significant use of ethenone is the synthesis of
Ketenes
', 2nd edition. John Wiley & Sons, 2006, .
organic chemistry
Organic chemistry is a subdiscipline within chemistry involving the scientific study of the structure, properties, and reactions of organic compounds and organic materials, i.e., matter in its various forms that contain carbon atoms.Clayden, ...
, ethenone is the formal name for ketene, an organic compound
In chemistry, organic compounds are generally any chemical compounds that contain carbon-hydrogen or carbon-carbon bonds. Due to carbon's ability to catenate (form chains with other carbon atoms), millions of organic compounds are known. T ...
with formula or . It is the simplest member of the ketene
In organic chemistry, a ketene is an organic compound of the form , where R and R' are two arbitrary monovalent chemical groups (or two separate substitution sites in the same molecule). The name may also refer to the specific compound ethen ...
class. It is an important reagent for acetylations.
Properties
Ethenone is a highly reactive gas (atstandard conditions
Standard temperature and pressure (STP) are standard sets of conditions for experimental measurements to be established to allow comparisons to be made between different sets of data. The most used standards are those of the International Union ...
) and has a sharp irritating odour. It is only reasonably stable at low temperatures (−80 °C). It must therefore always be prepared for each use and processed immediately, otherwise a dimerization to diketene
Diketene is an organic compound with the molecular formula , and which is sometimes written as . It is formed by dimerization of ketene, . Diketene is a member of the oxetane family. It is used as a reagent in organic chemistry. It is a colorles ...
occurs or it reacts to polymers that are difficult to handle. The polymer content formed during the preparation is reduced, for example, by adding sulfur dioxide to the ketene gas. Because of its cumulative double bonds, ethenone is highly reactive and reacts in an addition reaction H-acidic compounds to the corresponding acetic acid derivatives. It does for example react with water to acetic acid or with primary or secondary amines
In chemistry, amines (, ) are compounds and functional groups that contain a basic nitrogen atom with a lone pair. Amines are formally derivatives of ammonia (), wherein one or more hydrogen atoms have been replaced by a substituent such ...
to the corresponding acetamide
Acetamide (systematic name: ethanamide) is an organic compound with the formula CH3CONH2. It is the simplest amide derived from acetic acid. It finds some use as a plasticizer and as an industrial solvent. The related compound ''N'',''N''-dimet ...
s.
Preparation
In industrial chemistry, ketene is produced by thedehydration reaction
In chemistry, a dehydration reaction is a chemical reaction that involves the loss of water from the reacting molecule or ion. Dehydration reactions are common processes, the reverse of a hydration reaction.
Dehydration reactions in organic ch ...
of acetic acid:
: History
When passed through heated pipes or electrically heated metal (likecopper
Copper is a chemical element with the symbol Cu (from la, cuprum) and atomic number 29. It is a soft, malleable, and ductile metal with very high thermal and electrical conductivity. A freshly exposed surface of pure copper has a pinkis ...
) wires at 500-600 °C in the presence of carbon disulfide
Carbon disulfide (also spelled as carbon disulphide) is a neurotoxic, colorless, volatile liquid with the formula and structure . The compound is used frequently as a building block in organic chemistry as well as an industrial and chemical n ...
, acetone decomposes into methane
Methane ( , ) is a chemical compound with the chemical formula (one carbon atom bonded to four hydrogen atoms). It is a group-14 hydride, the simplest alkane, and the main constituent of natural gas. The relative abundance of methane on Ea ...
and ethenone, with 95% yield.
Ethenone was discovered at the same time by Hermann Staudinger (by reaction of bromoacetyl bromide with metallic zinc
Zinc is a chemical element with the symbol Zn and atomic number 30. Zinc is a slightly brittle metal at room temperature and has a shiny-greyish appearance when oxidation is removed. It is the first element in group 12 (IIB) of the periodi ...
)H. Staudinger H. W. Klever (1908): "Keten. Bemerkung zur Abhandlung zur Abhandlung der HHrn. V.T. Wilsmore und A. W. Stewart". ''Berichte der deutschen chemischen Gesellschaft'', volume 41, issue 1, pages 1516-1517. Tidwell, T. T. (2005), "Ein Jahrhundert Ketene (1905–2005): die Entdeckung einer vielseitigen Klasse reaktiver Intermediate". ''Angewandte Chemie'', volume 117, pages 5926–5933. The dehydration of acetic acid was reported in 1910.
 The thermal decomposition of acetic anhydride was also described.
The thermal decomposition of acetic anhydride was also described.



Natural occurrence
Ethenone has been observed to occur in space, in comets or in gas as part of the interstellar medium.Use
Ethenone is used to make acetic anhydride from acetic acid. Generally it is used for the acetylation of chemical compounds. : :
:sorbic acid
Sorbic acid, or 2,4-hexadienoic acid, is a natural organic compound used as a food preservative. It has the chemical formula and the structure . It is a colourless solid that is slightly soluble in water and sublimes readily. It was first isol ...
by reaction with 2-butenal
Crotonaldehyde is a chemical compound with the formula CH3CH=CHCHO. The compound is usually sold as a mixture of the ''E''- and ''Z''-isomers, which differ with respect to the relative position of the methyl and formyl groups. The ''E''-isomer ...
(crotonaldehyde) in toluene
Toluene (), also known as toluol (), is a substituted aromatic hydrocarbon. It is a colorless, water-insoluble liquid with the smell associated with paint thinners. It is a mono-substituted benzene derivative, consisting of a methyl group (CH3) a ...
at about 50 °C in the presence of zinc salts of long-chain carboxylic acids. This produces a polyester of 3-hydroxy-4-hexenoic acid, which is thermally or hydrolytically depolymerized to sorbic acid.
Ethenone is very reactive, tending to react with nucleophiles
In chemistry, a nucleophile is a chemical species that forms bonds by donating an electron pair. All molecules and ions with a free pair of electrons or at least one pi bond can act as nucleophiles. Because nucleophiles donate electrons, they are ...
to form an acetyl group. For example, it reacts with water to form acetic acid; with acetic acid to form acetic anhydride; with ammonia and amines to form ethanamides; and with dry hydrogen halides to form acetyl halides.
The formation of acetic acid likely occurs by an initial formation of 1,1-dihydroxyethene
1,1-Dihydroxyethene is an organic compound consisting of two hydroxy groups as substituents on the same carbon atom of an ethene chain. The chemical is also called ketene hydrate because it is the carbonyl hydrate of ketene. Its structure can als ...
, which then tautomerizes to give the final product.
Ethenone will also react with itself via + 2photocycloadditions to form cyclic dimers known as diketene
Diketene is an organic compound with the molecular formula , and which is sometimes written as . It is formed by dimerization of ketene, . Diketene is a member of the oxetane family. It is used as a reagent in organic chemistry. It is a colorles ...
s. For this reason, it should not be stored for long periods.
Hazards
Exposure to concentrated levels causes humans to experience irritation of body parts such as the eye,nose
A nose is a protuberance in vertebrates that houses the nostrils, or nares, which receive and expel air for respiration alongside the mouth. Behind the nose are the olfactory mucosa and the sinuses. Behind the nasal cavity, air next passes ...
, throat
In vertebrate anatomy, the throat is the front part of the neck, internally positioned in front of the vertebrae. It contains the pharynx and larynx. An important section of it is the epiglottis, separating the esophagus from the trachea (windpip ...
and lungs. Extended toxicity testing on mice, rats, guinea pigs and rabbits showed that ten-minute exposures to concentrations of freshly generated ethenone as low as 0.2 mg/liter (116 ppm) may produce a high percentage of deaths in small animals. These findings show ethenone is toxicologically identical to phosgene.
The formation of ketene in the pyrolysis
The pyrolysis (or devolatilization) process is the thermal decomposition of materials at elevated temperatures, often in an inert atmosphere. It involves a change of chemical composition. The word is coined from the Greek-derived elements ''py ...
of vitamin E acetate
A vitamin is an organic molecule (or a set of molecules closely related chemically, i.e. vitamers) that is an essential micronutrient that an organism needs in small quantities for the proper functioning of its metabolism. Essential nutrie ...
, an additive of some e-liquid
An electronic cigarette is a handheld battery-powered vaporizer that simulates smoking, but without tobacco combustion. E-cigarette components include a mouthpiece (drip tip), a cartridge (liquid storage area), a heating element/ atomizer, a mic ...
products, is one possible mechanism of the reported pulmonary damage caused by electronic cigarette use.
A number of patents describe the catalytic formation of ketene from carboxylic acids and acetates, using a variety of metals or ceramics, some of which are known to occur in e-cigarette devices from patients with e-cigarette or vaping product-use associated lung injury (EVALI).U.S. patent No. 5475144. Catalyst and process for synthesis of ketenes from carboxylic acids. Dec 12, 1995. https://patents.google.com/patent/US5475144A/en
Occupational exposure limits are set at 0.5 ppm (0.9 mg/m3) over an eight-hour time-weighted average.
An IDLH
The term immediately dangerous to life or health (IDLH) is defined by the US National Institute for Occupational Safety and Health (NIOSH) as exposure to airborne contaminants that is "likely to cause death or immediate or delayed permanent advers ...
limit is set at 5 ppm, as this is the lowest concentration productive of a clinically relevant physiologic response in humans.
References
Literature
* Tidwell, Thomas T.Ketenes
', 2nd edition. John Wiley & Sons, 2006, .
External links
* {{Chemical agents Ketenes Gases Pulmonary agents Acetylating agents